Water has many detrimental effects to the fuel. Dissolved water, for instance, can affect fuel stability, while water in its free and emulsified states can be more problematic to the engine fuel system. Not only can water cause damage to the fuel system, including the sensitive fuel injectors, it also promotes microbial growth within a fuel tank. When enough water is present, being that it is heavier than diesel, it will fall to the bottom of a fuel tank where additional acids can be formed, causing damage to tank walls and bottoms. A layer also forms between the diesel and free water boundary and this is where microbial contaminants will grow, feeding off the hydrocarbon, but living within the water.
Dissolved Water
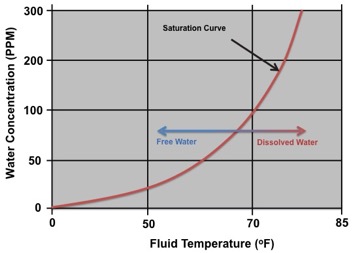
All diesel fuels have the ability to retain water within the fuel in a dissolved state. The ability of the fuel to hold the water in a dissolved state is highly dependent on the temperature of the fuel and its overall condition. Much like a mug of hot coffee can retain a certain volume of sugar dissolved within the water, it is the same for water in fuel. Taking the mug of hot coffee and sugar as an example, the coffee will dissolve the sugar and hold it chemically within the water while it remains hot. Sugar can continue to be added and dissolved, however, at some stage the hot coffee will be saturated with sugar and no longer be able to hold any more within solution. At this point, sugar will start to collect at the bottom of the mug, instead of dissolving into the coffee. This is what is known as the Saturation Point. Additionally, as the mug of coffee cools down over time, more and more sugar will fall out of solution and down to the bottom of the mug. This phenomenon is identical to that with water contamination in diesel fuel.
All diesel fuels have the ability to retain water in a dissolved state up to a certain volume. With most #2 diesel fuels found around the world, it is typically around 140-240ppm (0.014 – 0.024% by volume). In fact, many of the world’s diesel engine OEM’s clearly specify that water contamination must remain below 200ppm to ensure the reliable operation of the fuel injection system. The image below illustrates a typical saturation curve for a diesel fuel. As can be seen, the ability for the fuel to retain water in a dissolved state is highly dependent on temperature.

Emulsified Water
This is the state whereby microscopic water droplets have fallen out of a dissolved state, and due to the turbulence and the mixing effects within an operating diesel system, the water becomes emulsified and is unable to fall away and to the bottom of the tank. The images below show diesel fuel with water contamination in both a free and emulsified state. Note that the water contamination within the examples has been dyed blue to provide a more clear representation of the 2 states.


Free Water
Free water is water that falls out of suspension in the fuel and gathers at the bottom of the fuel tank, as seen below. This can only occur once the diesel fuel has been saturated with water in a dissolved state and the temperature conditions are not high enough to hold additional fuel.

Diesel fuel that is found to be in a hazy condition is typically found to be contaminated with water to some degree held in a dissolved state. Diesel fuels should be continually monitored for water contamination levels whilst in storage and maintained at levels below 200ppm.
Microbial Growth
There is one simple rule when it comes to microbial growth in a diesel fuel tank; it cannot exist without the presence of water. Many fuel consumers, however, never actually treat the cause of the problem, but rather treat the symptom of the problem. There are literally thousands of products and brands on the market all offering their own unique benefits to remove and prevent microbial growth. The ONLY method, which actually guarantees the prevention of microbial growth, is to remove the catalyst (water) and prevent the contamination in the first place.
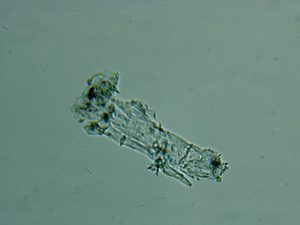
Asphaltenes are a bituminous material found in most diesel fuels with the concentration varying between batches and geographic location around the world. They are a natural contaminant from the refining process of producing various fuels out of a base crude oil. Asphaltenes are not soluble in fuel and are generally less than 2μm in size in their natural state. However, within diesel fuel they have a tendency to agglomerate, forming clusters in the order of 100-200μm, making them a key component to premature fuel filter blockage. Asphaltenes are NOT always filtered out by fuel producers in the refining process due to their small size and the expense of removal.
In some countries in Continental Africa and South America, emissions standards are less stringent and fuel sulfur levels are higher. In these locations, diesel sulfur levels remain at 500-5000ppm and there is some evidence to suggest that such fuels are manufactured with blends of light cycle oils from the refining process in order to use more from each barrel of crude oil. Fuels in these locations are generally black in color and contain large quantities of asphaltenes which makes the purification extremely challenging, and in many cases, very expensive.
It is a fact that most filter elements in use for diesel fuel applications are changed often due to the effects of asphaltenes. Filter elements that are coated by a black, tar-like substance have typically been affected by asphaltene contamination.